Applications and FAQs
How to Use CellCover

The Applications
A unique and special feature of CellCover liquid freezing is that you can perform additional molecular analysis on cells already analysed on morphological or molecular level: DNA (for e.g. PCR), RNA (for e.g RNA seq) or protein (for e.g protein modification analysis) can be isolated for subsequent applications after initial IHC, ICC or flow cytometry analyses.
These unique properties now enable new types of analyses of cellular functions. With CellCover liquid freezing, subpopulations of cells can be separated according to known properties by flow cytometry and subsequently analysed for gene expression profiles on RNA or on protein-level. Thus, with CellCover liquid freezing it is easy to link transcriptome and proteome of a given cell population.
Depending on your sample and experimental needs, you can choose the CellCover protocol that works best for you. Please scroll down for the protocols and FAQs.
The Opportunities
In comparison to commonly used molecular biological methods, CellCover adds following new analytical tools to bio-molecular research:
- Stabilize RNA and proteins simultaneously for several (2-3) days, without doubt of molecule integrity
- Cells maintain close to in vivo morphology without chemical crosslinking of biomolecules allowing straight forward RNA isolation.
- No freezing necessary
- Reliable analysis HMW-RNA and low stability enzymes even after day of storage
- Apply immunohistochemistry on formalin sensitive epitopes
- Apply in situ hybridization for analysis of gene expression
- Store cells conveniently for several days without change of protein or RNA expression profile or change in cells morphology as found with methanol
- Isolate subpopulations by flow cytometry based on protein labelling and perform subsequent transcriptome and/or proteome (MALDI) analyses.
- Next generation sequencing and single cell RNA seq.
- Reliable sample collection for biobank deposits.
Basic Protocols
Basic Protocol 1
Basic protocol for adherent cultured cells:
- Seed and grow cells on chamber slides
- Remove medium
- Wash cells 1x with PBS or CellCover
- Place slide in CellCover and store at 4°C until use
- Proceed to staining protocol according to experimental design, e.g. immunostaining (If RNA is to be isolated for downstream application, you can stain cells by using CellCover)
Basic Protocol 2
Basic immuno-staining protocol for suspension cells:
- Harvest cells by centrifugation
- Remove supernatant
- Flick tube to suspend cells in residual medium
- Add CellCover (5 to 10x volumes) and store at 4°C until use
- Pellet cells and remove SN
(Some cells might need higher centrifugation speed for pelleting.) - Proceed to standard staining protocols, e.g. immunostaining (If RNA is to be isolated for downstream application, you can stain cells by using CellCover as antibody diluent and washing buffer
Basic Protocol 3
Basic protocol for tissue specimens:
- Cut tissue into approximately 5 mm pieces
- CellCover does not penetrate tight junctions, thus encapsulated organs must be open across the largest diameter.
- Place tissue in CellCover (at least 10x volumes) and store at 4°C until use
- Change CellCover at least once after 4-24 hours
- Proceed according to experimental design e.g. RNA isolation

Special Protocol 1
Use CellCover prior to a protocol for cell dissociation (trypsinization protocol using trypsin EDTA) for effective cell fixation with no lysis of cells (including for single cell suspension).
Special protocol for adherent cultured cells (same protocol for single suspension):
- Remove cell culture media
- Add appropriate volume of CellCover and incubate for 2-3 min at room temperature
- Remove CellCover
- Add trypsin solution (e.g. Trypsin/EDTA 0,25% for adherent cells) and incubate for 5 min at 37°C (or until single cells detached)
- Collect cells by centrifugation
- Resuspend cells in CellCover and store at 4°C until needed or proceed with your (downstream) application(s)
For interest and quotes, please contact us at:
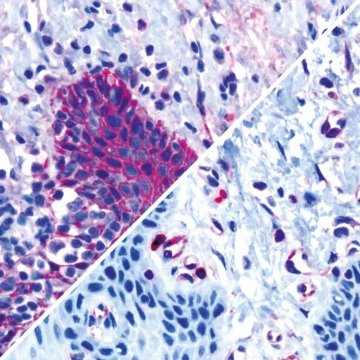
(Left: CK Pan, Right: CD31)
Immunohistochemistry
CellCover stabilizes cells in suspension, biopsies or tissue. CellCover is compatible with morphological analyses and staining procedures, with immunocytochemistry and flow cytometry.
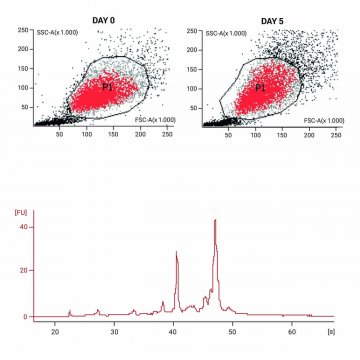
(Initial protein analysis & RNA isolation)
Double Analyses
Cells treated with CellCover maintain morphology for several days. Epitopes are protected and can be stained later. Highly intact RNA can be isolated from cells in which protein has already been stained by immunolabeling. Quality of RNA after CellCover protection: Cells have been immunostained for protein, RNA was isolated subsequently.
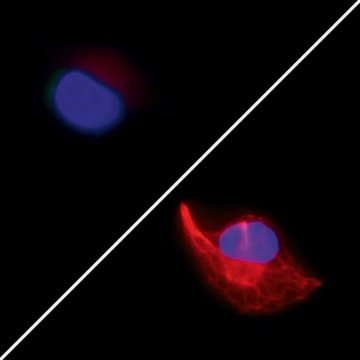
(Left: Formalin, Right: CellCover)
Immunohistochemistry
CellCover liquid freeze effect allows visualization of formalin sensitive epitopes like Vimentin, enabling new insights in cells structure and architecture.
Possible downstream applications:
Application of CellCover is simple: Remove cell culture medium, apply CellCover and incubate for two minutes. Then proceed with your standard protocol.
CellCover allows for many downstream experiments by avoiding lysis of cells at the start of sample preservation with complete cell fixation. By preventing lysed cells, cell morphology is preserved, allowing for accurate visualization of molecule location.
A unique and special feature of CellCover is the ability to perform additional molecular analysis on cells that have already been analyzed on a morphological or molecular level. DNA, RNA, or proteins can be isolated for example for NGS, RNA purification, or protein expression and proteomic analysis after initial immunohistochemistry, immunocytochemistry, or flow cytometry analyses.
A non-comprehensive list of compatible techniques are listed below:
- Batch and single cell analysis
- Flow cytometry / FACS
- Immunocytochemistry
- Immunohistochemistry
- FISH
- Microarray
- NGS
- PCR
- RNA Sequencing
- Northern Blotting
- Western Blotting
- Many more applications
For interest and quotes and for further questions concerning our product and applications, please contact us at:
FAQs
Can I store my purified RNA in CellCover?
Yes, you can. Storage of RNA in CellCoverprotects your RNA sufficiently for most subsequent purposes.
How shall I store my purified RNA in CellCover?
This really depends on when do you need it. After purification you usually have it on ice. If you need it for e. g. a Northern analysis within the next two days, keep it on ice (best in a cold room for the ice lasts longer). If you are not sure when you start your subsequent analysis, freeze it at minus 20°C. However, if you want proper maintenance for several month consider storage at -80°C.
My purified RNA is partly degraded, what might have been wrong?
Degradation, usually from the 5´end, is a common problem when using purification kits. We recommend to strictly adhere to instructions of the manufacturer of the kit and to work as cold as possible (above 0°C). After precipitation, we recommend dissolving purified RNA in ice-cold CellCover.
Does my purified RNA degrade in CellCover?
RNA protection by CellCoveris almost as effective as deep freezing and sufficient for most purposes (e.g. no change in RIN after 24 h on ice).
What are typical RIN values if using CellCover?
RIN values using CellCover are generally among the best you can get. You can expect RIN values of 10 even after storing cells for days in a fridge. However, many variables influence the individual experiment, e.g. RIN value increases the more experienced the researcher is, is dependent on the kit used for purification and might differ from cell type to cell type.
In tissue, how long does it take until my RNA is protected with CellCover?
CellCover works on to ends, chemical stabilisation and protection against enzymatic degradation. Time to fully protect RNA increases in the following order: non-adherent cells- adherent cells – spheroids – tissue. As a guideline: the more cells, the longer it takes. Usually storing a 5 mm tissue block sample on ice o/n is sufficient. Store spleen or central nervous system tissue for at least 24 h on ice.
Can I freeze my tissue samples in CellCover?
You can freeze your sample in CellCover, if protein in your sample is of interest for you. However, you should leave your sample on ice o/n for some time before freezing.
Is there something important to know when isolating RNA from tissue in CellCover?
Any manipulation might affect your RNA. Cell lysis is one of the most critical steps for it must be fast and efficient. Cells can be resuspended in a small volume CellCover before adding Lysis Buffer. Tissue should be minced on ice or grind under liquid nitrogen before adding Lysis Buffer.
Can I analyse my cells in CellCover with MALDI?
Yes, you can. Follow your lab protocol. Ingredients of CellCover do not interfere with macromolecular analyses.
Does CellCover protect HMW-RNA?
CellCover protects5´prime ends of RNA , so you can analyse large RNA species more easily. Some 20 kbases should not be a problem.


